On Oct. 14, Forklift Danceworks will present WesWorks, a performance that celebrates the skilled movement and the often unheard stories of the people whose work sustains the daily lives of the Wesleyan campus. In this Q&A we speak with Penny Snyder '16, who works as the communications manager for Forklift. At Wesleyan, she majored in English and received High Honors for her general scholarship thesis on art museums, architecture, and public space. She is an incoming graduate student at the Lyndon B. Johnston School of Public Affairs at the University of Texas at Austin. Q: Hello Penny! How were you…
For a truly sustainable future, Class of 2016 alumni Kotaro Aoki and Kota Uno believe it's crucial to teach people how to view—and properly "use"— nature. "Education is the most important piece in solving the root cause of climate change and environmental problems," Aoki said. "If we don't change our mindset, the same problem continues to rise no matter how drastic changes on the surface are." After reuniting recently in Fukushima, Japan during the COVID-19 pandemic, Aoki, a philosophy major, and Uno, a College of Social Studies major, discovered a shared interest in climate change. They agreed that they needed…
Johan "Joop" Varekamp, Harold T. Stearns Professor in Earth Science, professor of earth and environmental studies, is the co-author of an article published in Geology, March 2021. The study, titled "Volcanic Carbon Cycling in East Lake, Newberry Volcano, Oregon," focuses on the bubbling East Lake, the site of the Newberry Volcano, and the geological implications of the carbon reactions happening there. Varekamp co-authored the article with graduate student Christina Cauley and former students: Hilary Brumberg '17, Lena Capece '16, Celeste Smith '19, Paula Tartell '18, and Molly Wagner MA '19. The team researched this geological phenomenon from 2015 to 2019,…
On March 18, the Center for the Arts presented "A Conversation with Theater Artist Miranda Haymon ’16." Haymon, visiting instructor of theater, is Wesleyan's inaugural Breaking New Ground Theater Artist-in-Residence, a new residency that brings early-career Black, Indigenous, People of Color (BIPOC) theater artists to campus. The discussion was led by Sam Morreale ’19. During the conversation, Haymon discussed artistic processes, Blackness, queerness, Brechtian analysis, the impacts of the pandemic on artmaking, and ideas for the future. Haymon compared a theater performance to a "living document" in which the performance, audience, and actors are constantly changing. "The work changes, and…
Wesleyan in the News 1. The Chronicle of Higher Education: "Beware the Doyens of Disruption" In this op-ed, President Michael Roth '78 responds to predictions that COVID-19 is going to "change everything" in higher education with a reminder that "the desire of bright young people from all over the world for an on-campus education remains strong." He writes, "It’s because the connectivity among people and practices that takes place in person intensifies the learning experience." 2. HxA Podcast: "Michael Roth, Safe Enough Spaces" President Michael Roth '78 is interviewed on the Heterodox Academy's podcast about his book, Safe Enough Spaces:…
The Department of State’s Bureau of Educational and Cultural Affairs (ECA) recently announced that Wesleyan is included on the list of United States colleges and universities that produced the most 2019–2020 Fulbright U.S. Students. The Fulbright Program is the U.S. government's flagship international educational exchange program. Not only is Wesleyan a top Fulbright producer nationwide with its seven grantees, but it also has more winners than any other liberal arts institution in Connecticut. “We are delighted to see that the colleges and universities we are honoring as 2019–2020 Fulbright top-producing institutions reflect the geographic and institutional diversity of higher education…
On May 8, the Office of Student Affairs hosted a reception honoring students who received academic or leadership prizes, fellowships, and scholarships in 2018–19. More than 315 students and recent alumni received one of the University's 180 prizes. (View the list below or on the Student Affairs website.) Scholarships, fellowships, and leadership prizes are granted to students and student organizations based on criteria established for each prize or award. Certain University prizes are administered by the Student Affairs/Deans’ Office, while others are administered by the Office of Student Activities and Leadership Development (SALD). (more…)
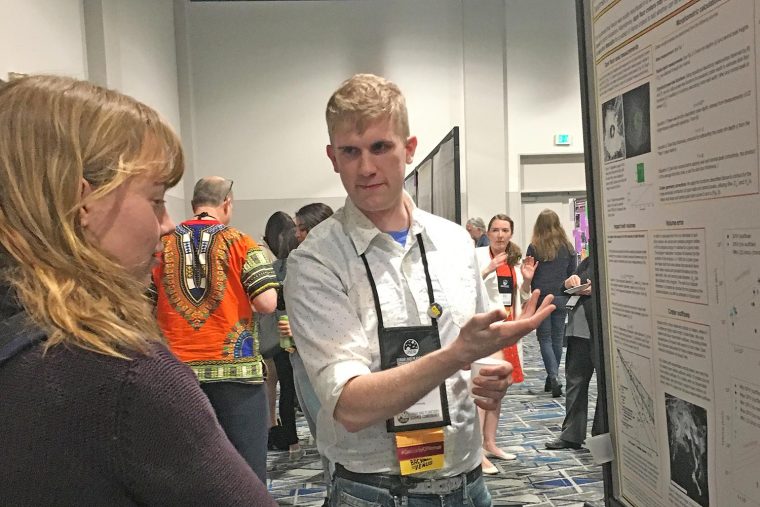
Several Wesleyan students, faculty, and alumni attended the 50th Lunar and Planetary Science Conference (LPSC) March 18-22 in The Woodlands, Texas. Members of the Wesleyan Planetary Sciences Group presented their research on a range of planetary bodies. This annual conference brings together international specialists in petrology, geochemistry, geophysics, geology, and astronomy to present the latest results of research in planetary science. Earth and environmental studies major Emmy Hughes '20 presented a poster titled “Observations of Transverse Aeolian Ridges in Digital Terrain Models" during a session on “Planetary Aeolian Processes.” Earth and environmental science graduate student Reid Perkins MA '19 presented a…
Sophie Zinser ’16, deputy director of Syria Direct in Amman, has been selected as a Schwarzman Scholar, one of the world’s most prestigious graduate fellowships, located at Schwarzman College at Tsinghua University in Beijing. This Schwarzman Class of 2020 is only the fourth in the program’s history, with 147 Schwarzman Scholars selected from over 2,800 applicants. The class is comprised of students from 38 countries and 119 universities, with 40 percent originating from the United States, 20 percent from China, and 40 percent from the rest of the world. The Class of 2020 will enroll in August 2019. In a…
Ellen Paik ’16, an analyst at Goldman Sachs, teamed up with three colleagues to pitch New Story, an organization working on developing low-cost housing solutions via 3D printing technology, to Goldman Sachs’ CEO and Partnership Committee as part of the Analyst Impact Fund, a global firmwide competition. The prize: a grant to the finalist teams’ selected nonprofits. The event was broadcasted live online on Yahoo Finance (see Paik's team come in around 38 minutes). Paik’s group placed second in the global finals and earned New Story $75,000 in support of the organization’s 3D printing initiative. The grant will go towards building…
Melissa Leung '16 is 1 of 75 Americans selected to participate in the 2018–2019 Congress-Bundestag Youth Exchange (CBYX) for Young Professionals, a yearlong fellowship for study and work in Germany. CBYX for Young Professionals provides opportunities for youth to collaborate, interact with new people and new ideas, and, ultimately, to become better global citizens and better leaders. The program annually provides scholarships to 350 Americans and also brings 360 Germans to the United States. While in Germany, Leung will attend a two-month intensive German language course, study at a German university or professional school for four months, and complete a…
Faculty, graduate students, and alumni attended the 49th Lunar and Planetary Science Conference March 19–23 in The Woodlands, Texas. Three graduate students were awarded funds from the NASA Connecticut Space Grant that allowed them to travel to this meeting. Earth and environmental sciences graduate student Reid Perkins presented a research poster titled "Where Are the Missing Tessera Craters on Venus?" Perkins's advisor is Martha Gilmore, the George I. Seney Professor of Geology, professor of earth and environmental sciences. Earth and environmental sciences graduate student Melissa Luna presented a poster titled "Multivariate Spectral Analysis of CRISM Data to Characterize the Composition of Mawrth…